|
<NOTE>
Chimpanzee Pith-Folding at Toro-Semliki Wildlife Reserve, Uganda
William C. McGrew1 & Kevin D. Hunt2
1 Department of Archaeology & Anthropology, University of Cambridge, UK 2 Department of Anthropology, Indiana University, USA INTRODUCTION Chimpanzees daily make and use tools, and most of their tool manufacturing transforms vegetation by stripping, peeling, splitting, crushing, clipping, etc. In habituated populations, behavioural data accompany the artefacts, so that observers see precisely how the tools are made and why. However, chimpanzees also modify detached vegetation when not making tools, and these activities may leave behind puzzling artefacts. Such are the ‘wadges’ (or ‘quid’) of the pith of the wild date palm, Phoenix reclinata, which we analyse in detail here. The artefact is obvious when encountered: A straight stem is bent repeatedly to alternating sides, concertina-style, so that it has a series of folds at acute angles (see Figure 1). These objects are the spat-out products of buccal compression, from which juices have been extracted by squeezing them between tongue and palate. Their ‘function’ is straight-forwardly nutritional, but the puzzles exist: Why do they take this distinctive shape, and why do so many wadges have an odd number of folds while so few have an even number of folds? 
Figure 1. Folded pith wadges of Phoenix reclinata made by chimpanzees at Toro-Semliki Wildlife Reserve. The only previous report of leaf-folding comes from the chimpanzees of Bossou, Guinea, who manufacture water-extracting tools1–4. To make these tools, the apes fold sets of 1–4 leaves (mostly of Hybophrynium braunianum) at about 3-cm intervals, while stuffing them into the mouth. This device is inserted by hand into a tree-hole containing water, then extracted and sucked, as a source of sustenance. (Such leaf-folding differs from leaf-sponging and leaf-spooning, although all three techniques yield drinking water3.) Wadging entails a nutritious object being manipulated by mouth but not swallowed; it seems to be a universal chimpanzee food-processing technique. Goodall5 (p. 238) described wadging at Gombe in detail, noting a variety of food-items, mostly fibrous plant-parts, e.g. figs, bark, etc. In some cases, chimpanzees add leaves to fleshy, ‘rich’-tasting foods, such as meat, eggs, honey, or overripe fruit, to form a compressed, homogenised bolus. This may be swallowed, but usually there is an accumulation of amorphous, jumbled fibre-spheres left on the ground. (Presumably, wadges are ejected rather than ingested, because the fibrous mass of foliage is of low quality and would take up valuable gut space.) We have found no previous record of accordion-folded wadges from any other species of non-human primate. Here we document these special wadges and seek to infer how the artefacts end up with a non-random design, that is, a prevalence of odd-numbered folds. We predicted that the number of folds is positively correlated with the length of the pithy stem involved; given a standard unit of folding distance, the longer the stem, the more folds required. However, we had no clue a priori about the biased number of folds. METHODS We studied the wild chimpanzees (Pan troglodytes schweinfurthii) of the Toro-Semliki Wildlife Reserve, Uganda6. If the partly-habituated chimpanzees could not be un-nested at the beginning of the day, we searched for them, listening for calls and looking for their signs. Once contacted, we stayed with them for as long as possible. These apes occupy a largely open habitat, which is a mosaic of grassland, scrub, open woodland, gallery forest, and swamp. In tracking or following chimpanzees, we noted feeding traces left by them. For wild date palm wadges, we counted the numbers in an assemblage and noted the sources from whence the piths had been detached. Intact and fresh (less than 24 hr old) wadges were collected in plastic ziplock bags and returned to camp for processing. In camp, WCM measured wadges to the nearest 0.5 cm, from fold to fold, and counted the number of folds. Thus, a folded-pith wadge with ‘n’ folds yielded ‘n + 1’ measureable segments. RESULTS Remnants of the chimpanzees’ wadging of wild date palm were found daily over May-September 2008. A typical assemblage consisted of several mature fronds lying on the ground less than 5 m from the presumed source plant. A source plant was identified by freshly-damaged ends of the still-intact fronds; these were readily apparent because white pith revealed by the damage contrasted with the green outer colour of the foliage. Detached fronds were split length-wise and clipped cross-wise, producing segments of 8–50 cm length. All of the outer epithelium of the segments was peeled away, leaving only the grainy, moist, fibrous pith in strips of less than 1 cm diameter. Few were unused or incomplete strips of pith, by comparison with more than 30 wadges left strewn on the ground (See Figure 1). These artefacts often were concentrated in areas of less than 50 cm diameter, sometimes in a loose pile, as if the wadger had sat still and worked through a set of wadges before moving on. Fresh wadges had a distinctive pale gold colour and were moist; older wadges turned white and shrank in size as they dried. Desiccated wadges also expanded, changing the angles of the folds from acute to obtuse, unless the fibres were tangled up with one another. How did we know that these artefacts were made by chimpanzees, if we had not seen them being made? Several times observers were within 5 m of chimpanzees wadging palm fibre in thick undergrowth; we glimpsed fragments of their behaviour and heard the distinctive sounds of fronds being detached. We then recovered fresh artefacts from the site only minutes later. Once, a chimpanzee carried fronds into a tree and wadged arboreally. Also, we often saw the chimpanzees wadging other vegetation, e.g. at least 4 species of tree-bark (but all of these wadges were the more typical jumbled balls). Many wild date palm wadges were recovered when we tracked chimpanzees, only minutes ahead of us. We found imprints of knuckle- and hand-prints in association with the wadges. No other animals at the study-site did such wadging, nor were there resident humans present to do so. We measured 110 wadges to the nearest 0.5 cm. These had 1–14 folds; 56 had 3 folds, making it the modal number. Fully extended, the wadges averaged 15.9 cm long (n = 110, range 8–50, median & mode = 17.5). The mean distance between folds (i.e. length of segment) was 3.8 cm (n = 563, range = 1.5–6.5, median = 3.5, mode = 3.0). There was a positive correlation (Spearman’s rho, n = 11, rs = 0.98, p < .001, two-tailed) between mean length of wadge and the number of its folds, suggesting that segment-length is relatively standardised (see Figure 2). 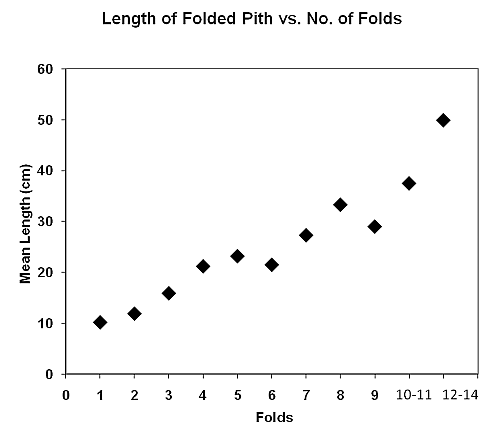
Figure 2. Average (overall) length of folded pith wadges of Phoenix reclinata as a function of number of folds. Many more wadges had an odd number of folds (1,3,5,7, etc.) than an even number of folds (2,4,6,8, etc). (See Figure 3) For every adjacent pairing, that is, 1 vs. 2 folds, 3 vs. 4 folds, etc., the prevalence of odd-number folds was greater. Presumably the predominance of 3–4 folds is a function of the preferred overall length divided by the typical segment length (see above). Overall, 91 (83%) wadges had 1–13 odd-numbered folds, while only 19 (17%) had 2–14 even-numbered folds (Binomial test, n = 110, z = 6.77, p < .001, two-tailed) (This was not a collection bias, as the number of folds was obscured in the field and could not be ascertained until the artefact was extended back in camp.) 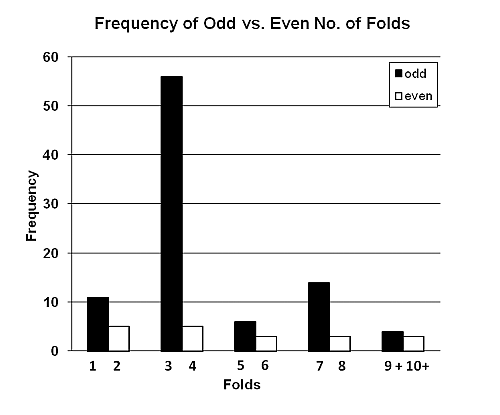
Figure 3. Frequency of folded pith wadges with odd versus even number of folds. DISCUSSION If the implicit interpretation of leaf-folding at Bossou is that the optimal dimension of the fold is a function of the space available in a chimpanzee’s mouth, then it makes sense for pith-folding too. The most economical way to pack a linear object into a much smaller space is to compact it to the maximum permissible length, and the most efficient form of compaction is folding. Thus it is notable that the average length of fold of leaves at Bossou was 3 cm versus 3.8 cm for pith-folding at Semliki. So, all other things being equal, the correlation between number of folds and total length of wadge is likely to be a matter of anatomical (buccal) constraints. (Other, leafy wadges are amorphous in shape, but the stiff longitudinal fibres of the pith make folding the better alternative.) But what about the odd-even difference? Perhaps chimpanzees consistently clipped lengths of pith that when folded to the optimal segment-length of about 3–4 cm were somehow biased toward an odd number of folds. This seems nonsensical. If an ape were sufficiently pernickety about producing exactly the right number of folds to fill up the mouth, then on average, all other things being equal, one would expect a 50:50 chance of odd or even, given individual variation in buccal volume. A solution to the oddness riddle emerged serendipitously from chance, unobstructed observations of habituated chimpanzees at another Ugandan field site, Kanyawara, in Kibale National Park. There, chimpanzees wadge the pith of papyrus (Cyperus papyrus) stems. They usually produce amorphous, jumbled-mass wadges, but they occasionally fashion concertina-shaped, folded wadges that are identical in form to the Semliki ones (Bertolani, pers. comm.; See Figure 4). The technique is as follows: After clipping the stem to the final length, the ape folds it in half, with one end in the lips and the other in one hand. She then ‘feeds’ the doubled-over stem into the mouth by the same hand, initial-fold first. Each new fold of the doubled-stem thus produces a pair of folds, which when added to the single initial fold, gives an odd-numbered total. Occasionally, the wadger does not bother with the initial fold, and just ‘feeds’ the stem into the mouth; presumably these artefacts have a 50:50 chance of ending up with an odd or even number of folds. 
Figure 4. Jumbled (standard) and folded pith wadges of Cyperus papyrus at Kanyawara, Kibale National Park. To what extent does pith-folding at Semliki resemble leaf-folding at Bossou? There are several differences3: The folded leaf is a tool, the pleats of which increase the water-holding ability of the leaves. The multi-folded pith is not a tool, and the folds seem to have no containing function. So, Semliki wadges are discarded after one use, whereas Bossou’s wadges may be re-used up to 122 times. At Semliki, only one species of plant is used for folding; at Bossou at least seven species are used. In making the wadge, at Semliki the raw material is folded over at its centre-point before being ‘fed’ into the mouth; at Bossou the raw material is stuffed directly into the mouth, starting at one end of the leaf. Most of these differences can be explained by the differing functions of the activities. On the other hand, there are some similarities: Both procedures modify plant materials in order to fill up the buccal cavity, using folding as a technique to do so. The resulting pleats are evenly and similarly spaced, yielding a concertina-like artefact with alternating folds. The making of both types of artefact involves hand-mouth coordination. Most of these similarities can be explained by the similar biomechanics of the tasks. Who cares if the number of folds in a wadge is odd or even? One answer is that this is the stuff of culture, not just of gross differences across populations, but also of nuanced variation, when cultural traits are basically similar but subtly different. Such trivial variants are common in human cultures, and the same may be true of non-human artefacts. This ethnographic note reminds us that culture is a layered phenomenon, and that if we operate on one level only, we may miss important features. Another answer is that researchers who work with artefacts, e.g. archaeologists, face challenges in inferring how those artefacts were produced. Etho-archaeology reminds us how difficult it can be to imagine the absent processes that result in material culture. AcknowledgementsWe thank: Uganda Wildlife Authority and Uganda National Council for Science and Technology for permission to work at Semliki, UWA rangers Alimosi Baluku, Patrick Biryomumsho, Charles Kasaija, Felix Anidraku, Justus Orobokiritto, and Elly Rutaro for assistance in the field; Moses Comeboy, Eriik Kasutama, Edson Katswamba, Jeremiah Nduthu for assistance in camp; Linda Marchant and Timothy Webster for research collaboration and comments on the manuscript; Lucie Salwiczek for graphics aid; Paco Bertolani for taking data at Kanyawara; an anonymous referee for helpful comments. References
Back to Contents |